When synthesizing aspirin in the lab, the product turns out to be a yellow solution and brown ppt when salicylic acid is reacted with acetic anhydride and H2SO4 (as a catalyst). What

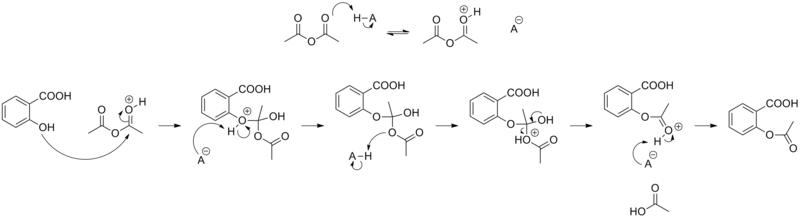
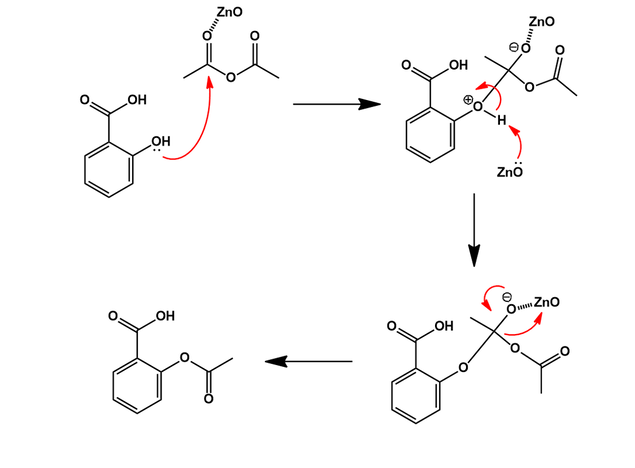
Draw the proper full electron-pushing mechanism for the reaction below, including all intermediates (with formal charges) and electron pushing arrow's. Label the electrophile and nucleophile in each s | Homework.Study.com
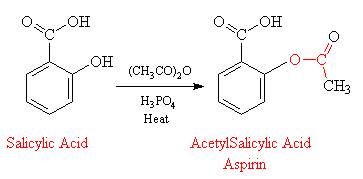
Pharmaceutical Chemistry Synthesis of Aspirin Principle : Synthesis of Aspirin from salicylic acid occurs by acetylating proce

What is the complete chemical mechanism for the base hydrolysis of aspirin to salicylic acid? | Homework.Study.com

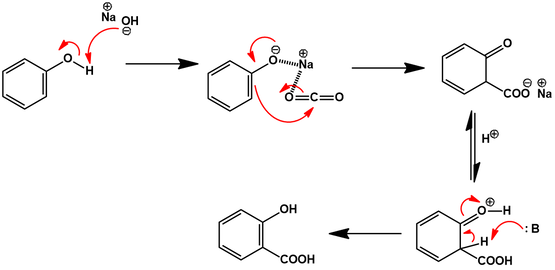
Illustrate the mechanism involved in the synthesis of aspirin without utilizing an acid catalyst. Although a metal catalyst is used in this green chemistry synthesis. The reactants involved in this reaction are
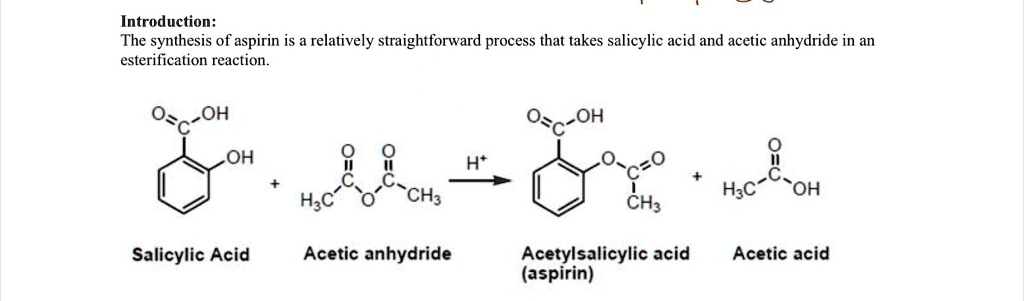
SOLVED: Draw a detailed mechanism of the synthesis reaction of aspirin. Include arrows ! Introduction: The synthesis of aspirin is a relatively straightforward process that takes salicylic acid and acetic anhydride in


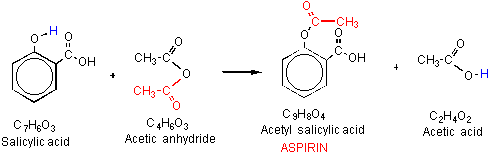

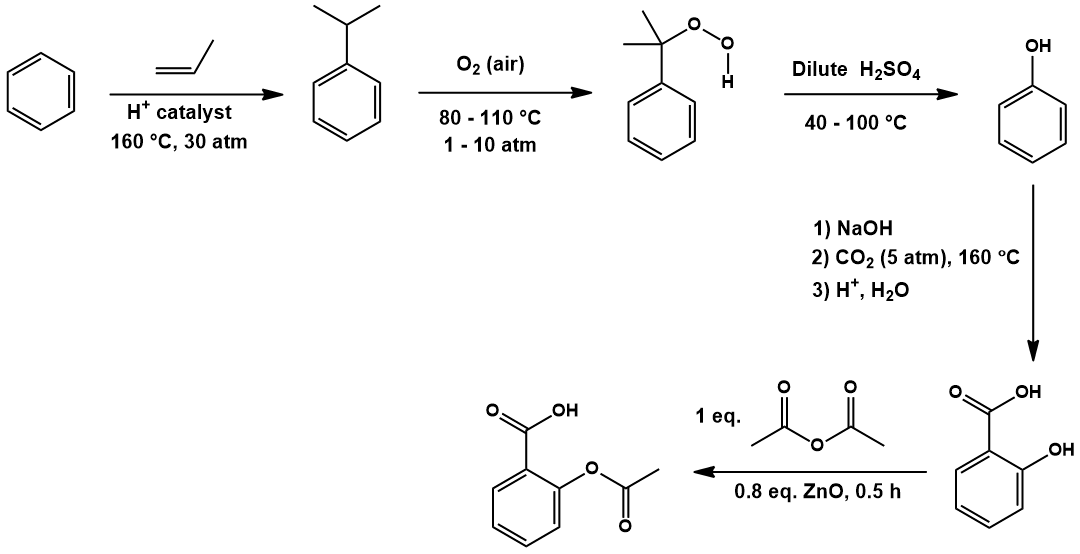
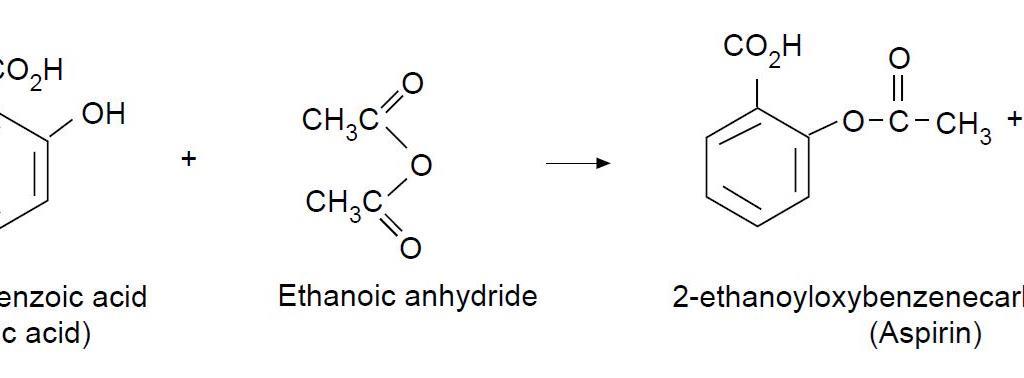
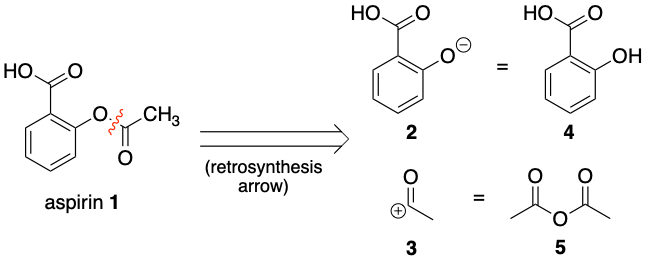
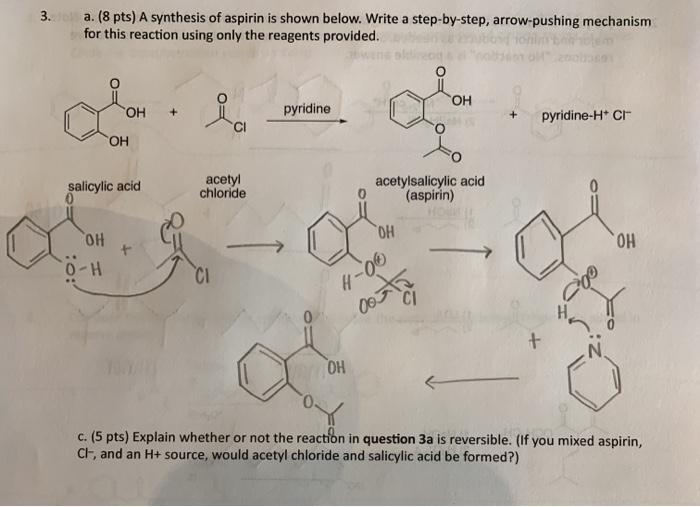
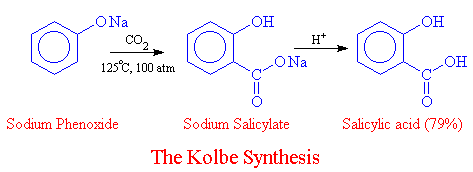
![PDF] The Synthesis and Analysis of Aspirin | Semantic Scholar PDF] The Synthesis and Analysis of Aspirin | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/5e9af12eecb0d610768b5c45b4f249cab0fbe630/5-Figure2-1.png)