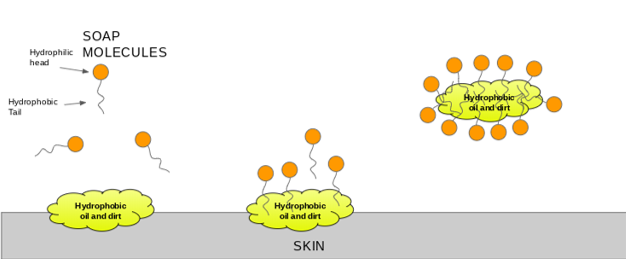
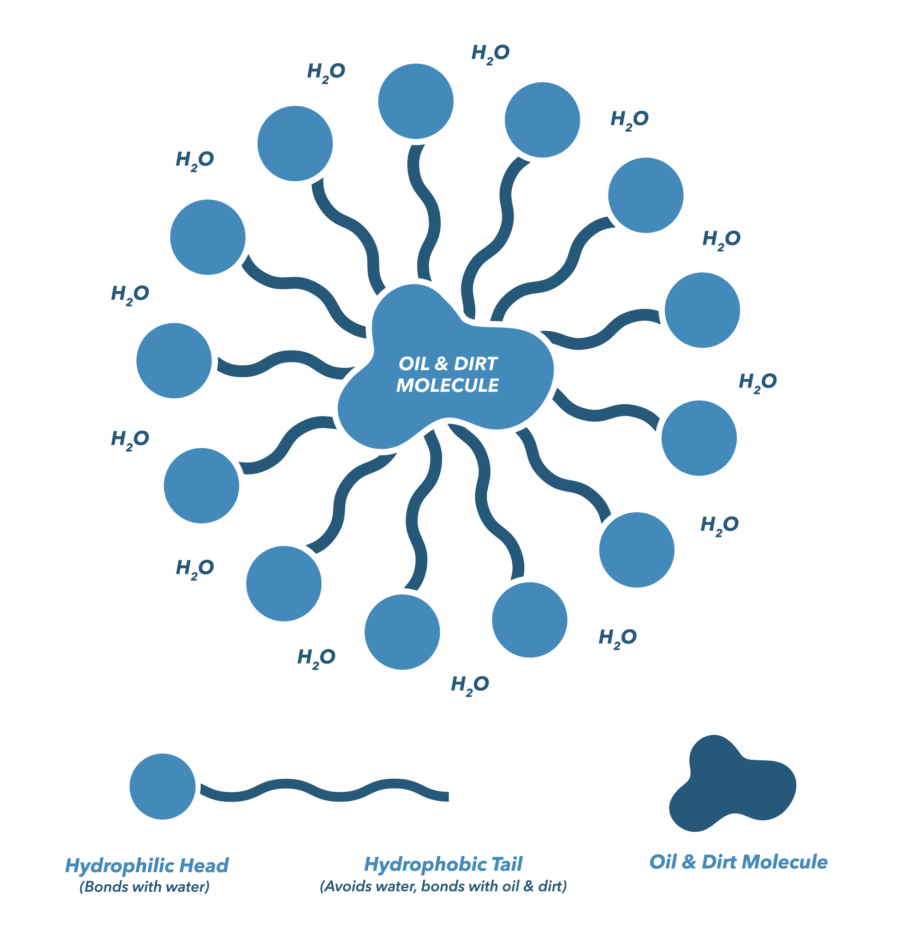
Explain micelle formation in soap solution. - Sarthaks eConnect | Largest Online Education Community
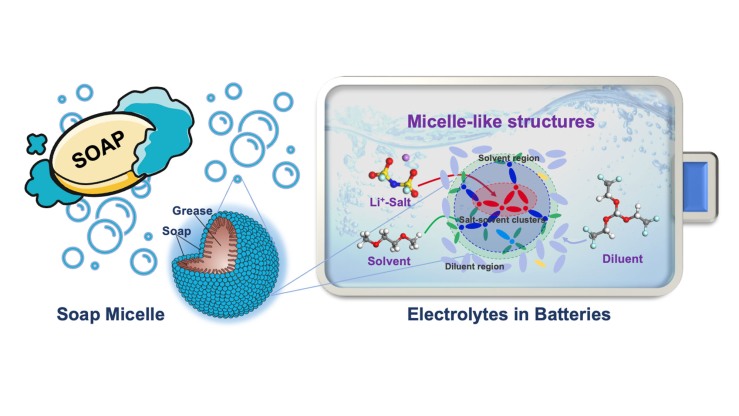
The secret to longer lasting batteries might be in how soap works, new study says | Brown University
![Draw neat labelled diagrams to show the hydrophobic and hydrophilic ends of a soap and micelle. [3 MARKS] Draw neat labelled diagrams to show the hydrophobic and hydrophilic ends of a soap and micelle. [3 MARKS]](https://search-static.byjusweb.com/question-images/byjus/infinitestudent-images/ckeditor_assets/pictures/21171/content_45544_2.jpg)
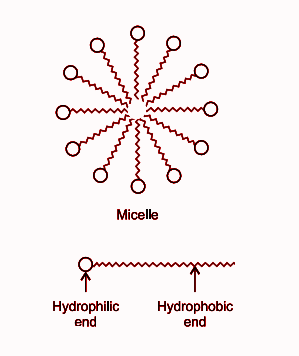
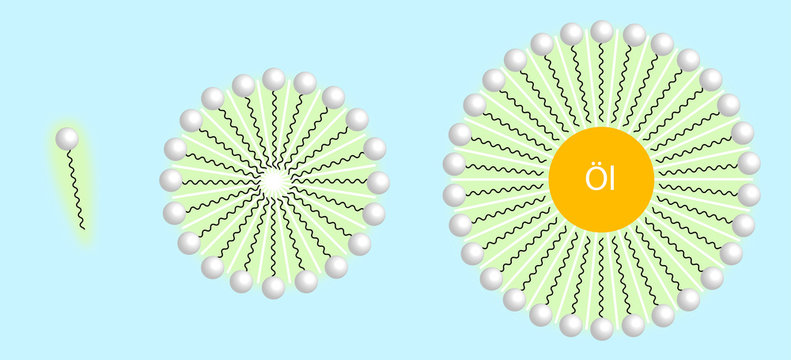
Draw neat labelled diagrams to show the hydrophobic and hydrophilic ends of a soap and micelle. [3 MARKS]


:max_bytes(150000):strip_icc()/soap-micelle-58ed36193df78cd3fcdf0908.jpg)




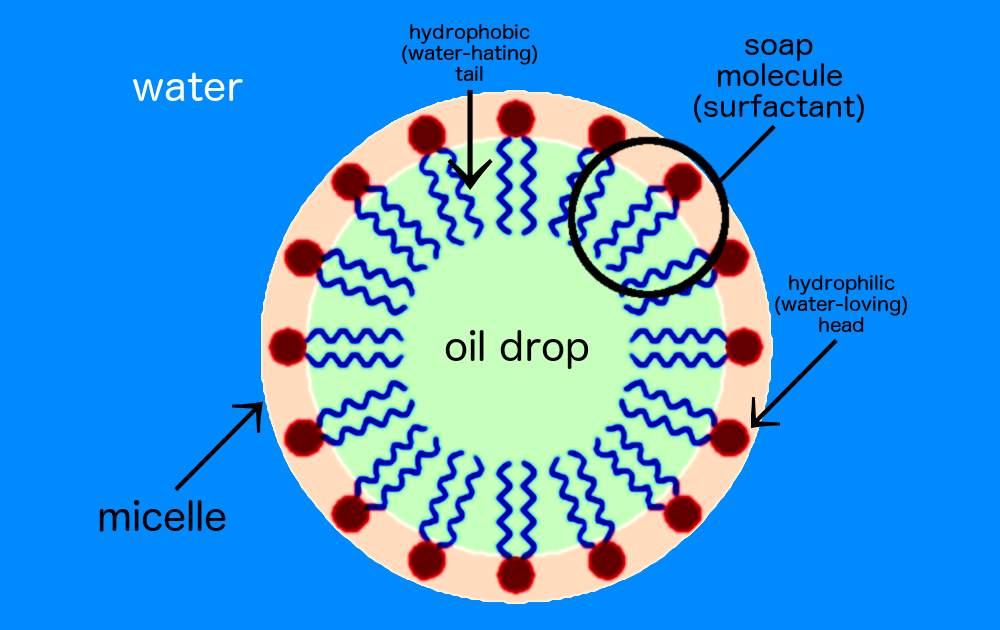
![Class 10 Carbon and its compounds] What are Micelles? - Teachoo Class 10 Carbon and its compounds] What are Micelles? - Teachoo](https://d1avenlh0i1xmr.cloudfront.net/c5536fd4-cc1f-4684-a708-ece69fada9ca/structure-of-micelle---teachoo.jpg)




![MCQ] In the soap micelles (a) the ionic end of soap is on the surface MCQ] In the soap micelles (a) the ionic end of soap is on the surface](https://d1avenlh0i1xmr.cloudfront.net/aed2074f-aafe-447a-b1f0-adb685bf01d5/structure-of-a-soap-micelle---teachoo.jpg)

