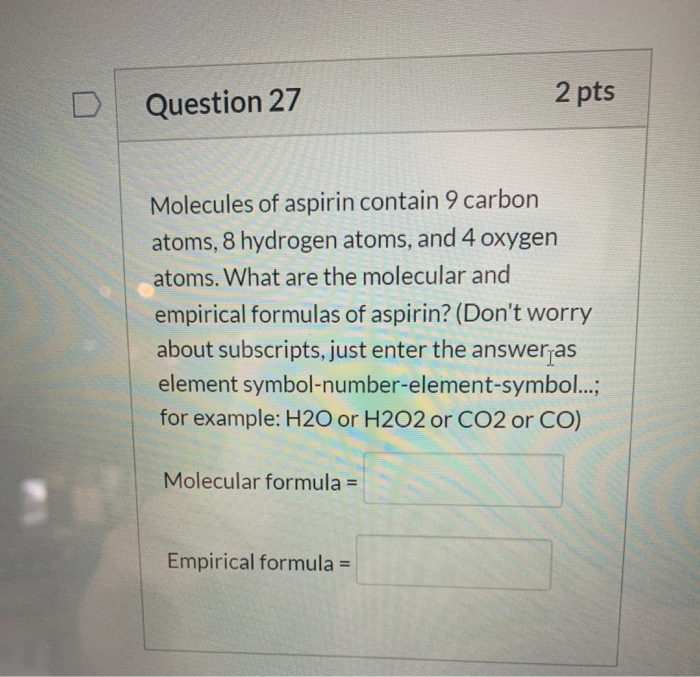
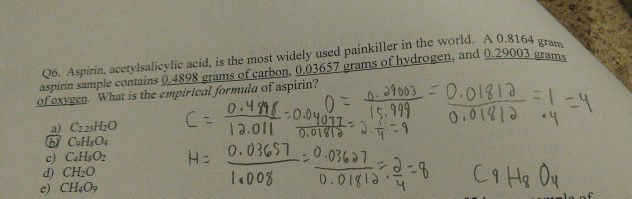SOLVED: MQueLA Aercke 17. Determine the molecular formula for the following compounds: Empirical formula is NO2; molecular mass is 84.04 g/mol. Empirical formula is CHCl; molecular mass is 148.44 g/mol. Empirical formula

64. Aspirin contain 60% C, 4.48% H and 35.5% O its empirical formula is a) C H20 (b) C,HO2 (c) C,H,04 (d) C H203

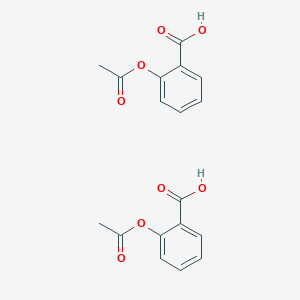
Write the structural formula for aspirin. Label the ester group and the carboxylic acid group. | Homework.Study.com





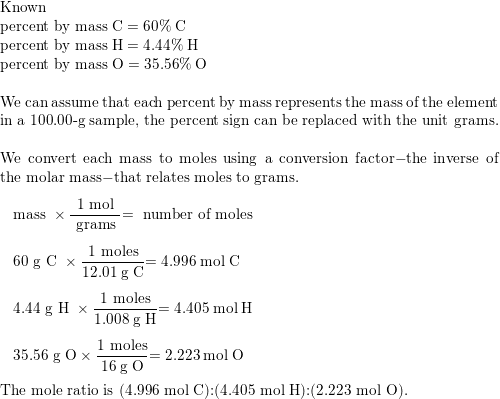


![Aspirin, formula and molecular structure.... - Stock Illustration [64032119] - PIXTA Aspirin, formula and molecular structure.... - Stock Illustration [64032119] - PIXTA](https://en.pimg.jp/064/032/119/1/64032119.jpg)